Work Packages
Work Package 1
Project management
- Project management
- Constant communication between the research team’s members.
- Prepare dissemination material and progress reports for the assessment of good scientific conduct and ethics
Work Package 2
Rational design of compounds (immunodominant peptides conjugated with mannosylated nanoparticles)
- Rational design of peptide analogues
- Selection of nanoparticle type for the encapsulation of peptides.
- Design of modified nanoparticles targeting the mannose receptor on Dendritic Cells (DCs).
The rational design of the compounds will be based on the following triptych:
- Selection of potent myelin epitope.
- Selection of nanoparticles.
- Surface modification of nanoparticles.
The modification in Lactic and glycolic acid copolymers [poly (lactic-co-glycolic) acid, PLGA] surface aims in conjugation with mannose units that could selectively deliver the NPs, along with the peptide-antigen, to the Mannose Receptors of the Dendritic Cells (DCs).DCs are the main antigen presenting cells involved in MS.
Work Package 3
Synthesis of peptides, development and analysis of modified nanoparticles
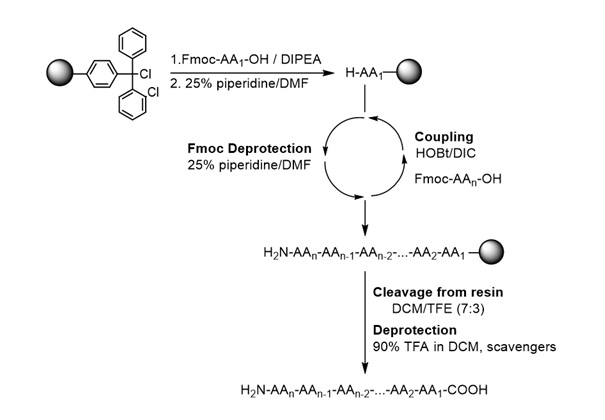
- Synthesis and identification of peptide analogues based on immunodominant myelin epitopes (Scheme 1).
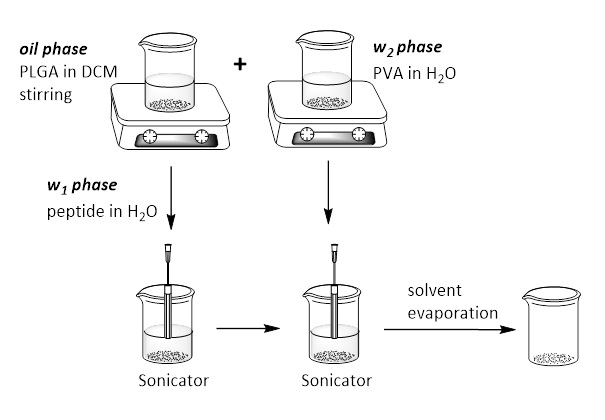
- Development of PLGA nanoparticles (identifications of their physicochemical characteristics) (Scheme 2).
- The synthesis of peptides will be achieved using the Fmoc/tBu methodology and 2-chlorotrytyl chloride resin.

- The development of PLGA [poly(lactic-co-glycolic) acid] nanoparticles, with peptides encapsulation will be carried out using double emulsion methodology with solvent evaporation, water-in-oil-in-water (w1/o/w2)

Phase w1, containing the peptide dissolved in water, is added in oil phase that contains the PLGA polymer dissolved in DCM. Two phases are emulsified with the use of a homogenizer and subsequently phase w2 containing the surfactant is added to produce the second emulsion. Finally, the emulsion is left (under fume hood or under pressure) for evaporation of solvents and final NPs forming.
Work Package 4
In vivo και in vitro αξιολόγηση συνθετικών μαννοζυλιωμένων νανοσωματιδίων – ανοσοκυρίαρχου πεπτιδίου της μυελίνης
- In vivo biological/immunological evaluation of synthesized molecules.
The in vivo evaluation will be carried out using Experimental Autoimmune Encephalomyelitis (EAE; C57BL/6 mice), animal model of Multiple Sclerosis (MS) by the supervision of Dr. Lesley Probert at Hellenic Pasteur Institute (Collaborating Organization).
All the in vitro / in vivo experimental protocols will be conducted in accordance with the Law “European Convention for the protection of animals used for experimental or other research purposes” and will be approved by the Institutional Committee for Animal care.
Work Package 5
Dissemination and communication management
- Dissemination, exploitation and assessment of the results to public
- The results of the project will be presented at national and international level with respect and observance of the principles of scientific ethics.
- The dissemination will take place through oral or poster presentations at scientific conferences and publications in peer-reviewed journals.
- Information material (webpage) with the results of the project aiming at business exploitation will be created.





